magazine_ Article
Pioneering biomedical research in South Tyrol
A new chapter for the CHRIS health study in Vinschgau/Val Venosta.
Ten years ago the unique Italian population study was initiated. Now CHRIS is entering its second phase, with 13,000 people taking part.
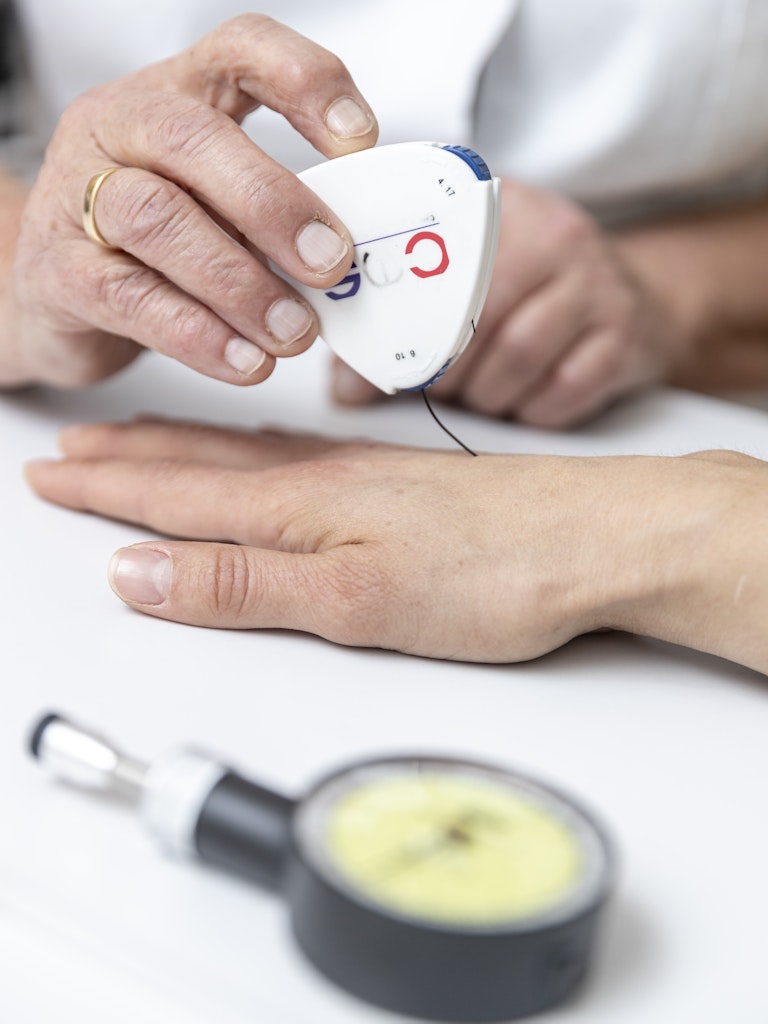
Recently, Martina from Schlanders/Silandro received an invitation to make an appointment at the CHRIS center in the local hospital for an interview, a blood test and a number of other health-related checks. She had already been there in 2011, as one of the first participants in the CHRIS population study. Following her initial assessment she decided to make appointments for her whole family. Since its beginnings ten years ago, the CHRIS study, conducted jointly by Eurac Research and the South Tyrol Health Authority, has become an increasingly important resource for regional and international research. With the start of the second phase, CHRIS is now becoming a prospective study in which it is possible to observe how the health status of the original participants have developed over time. In combination with genetic information, valuable new insights can be gained.
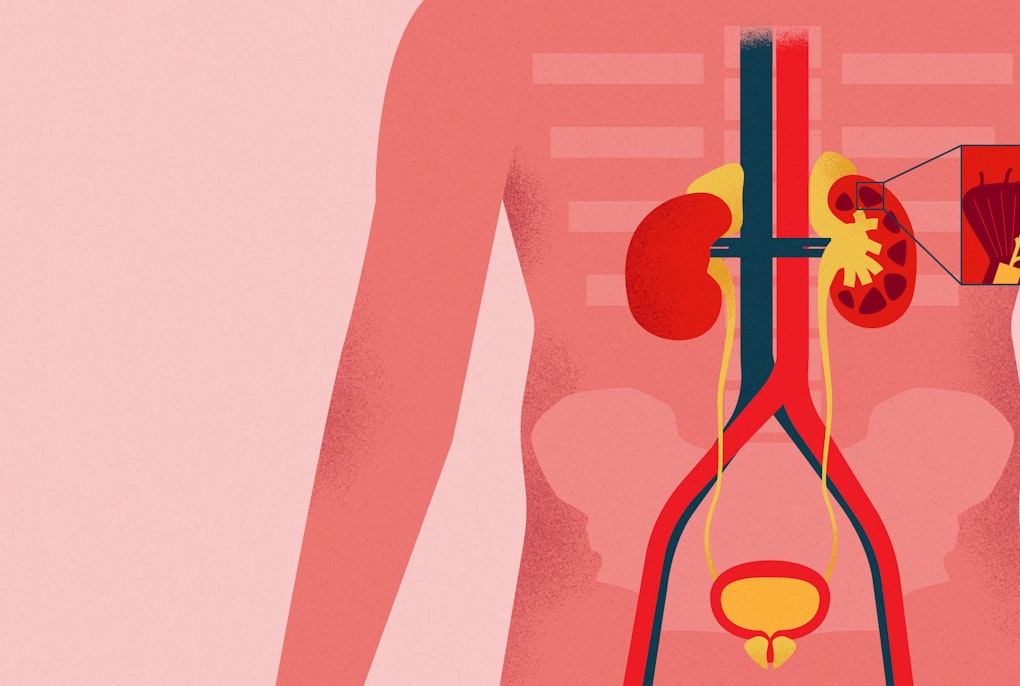
CHRIS is a cohort study - examining the health status of a specific group of people over time - is unique in its kind in Italy and worldwide. Since many families that permanently reside in the valley participate, the research teams can reconstruct genealogical information and observe the health status over several generations within the same family. Moreover, the group studied is representative of the general population, which is rare: often such studies only have the objective of researching a specific disease and are therefore limited to people suffering from that disease. A special feature of CHRIS is the close involvement of the population. In the communities of Upper Vinschgau/Val Venosta, more than a third of the population participated in the first study phase. Participation was also highly active in later specific sub-studies, for example on neurodegenerative diseases or fatty liver disease. Over the course of ten years, only one participant decided to drop out of the study. A wide range of data is collected from everyone who participates: Medical history and lifestyle information, a 20-minute electrocardiogram, more than a hundred biomarkers from blood and urine tests. The picture is further completed by the collaboration with the local Health Authority. With the consent of the persons concerned, the CHRIS database can merge the information from the study with existing Local Health Authority medical records. This system, although rather uncommon in Italy, has very comprehensive health data at its disposal gained through, and for research. The second phase will now decisively expand this information. All measurements will be repeated to observe the development of participant health over time. Only very few studies worldwide have been able to do this on such a broad data basis. The achievements in the past ten years of CHRIS are numerous. They include the biobank which was set up by the Institute of Biomedicine of Eurac Research in collaboration with the hospitals of Bozen/Bolzano and Meran/Merano. It is the largest of its kind in the province, storing one million biological samples at a temperature of -80 degrees Celsius and in compliance with the strictest safety standards. The research teams have also determined the genotype of the entire cohort and sequenced the exons in 3600 people, i.e. the most important gene segments that code for proteins. In 7000 people, they performed metabolomic analyses to study specific cellular processes. Thanks to this work and cooperation with other scientific institutions, the data from CHRIS has contributed to important research projects in Europe and around the world. In using CHRIS data, international studies have identified hundreds of genetic variants associated with kidney function, thyroid function, heart health, obesity, glucose metabolism and diabetes.
The CHRIS study also forms a valuable basis for the CHRIS-Covid-19 study, which has been underway since summer 2020. During the acute first phase of the pandemic, "regular" CHRIS study operations had to be suspended. and the CHRIS Covid-19 study was quickly launched. The subsidiary study now involves 4,500 CHRIS participants and their families. The goal of the study is to determine which genes cause severe covid-19 progression and, conversely, find out which genes protect against covid-19. The data and samples collected during the first study phase of CHRIS are now complemented by those from the Covid-19 study, forming a valuable resource that provides information on health status before and after possible SARS-CoV2 infection. At the same time, the CHRIS Covid-19 study helped monitoring the infection trends in Val Venosta/Vinschgau. In addition, as part of the world's largest genetics and Covid-19 research consortium (COVID-19 Host Genetics Initiative), the study data has also helped identify several locations in the genome that are associated with severe disease progression.
Population studies have always provided important medical insights and much knowledge we take for granted today was gained through them. It was the Framingham Study, which began in the U.S. in 1948, that first demonstrated the link between lifestyle and certain cardiovascular diseases, opening up possibilities for prevention. "The more information we gather, the more important CHRIS will become over time. Research technologies are constantly evolving. In genetic research, for example, the amount of data we can now analyze at affordable costs has reached unprecedented proportions and continues to grow. Thanks to modern biobanks and comprehensive databases of genetic information, we are moving ever closer to so-called precision medicine, in which prevention, diagnosis and treatment are tailored to the genetic and biological characteristics of the individual. In the field of oncology, this approach is already a reality, and it has the potential to transform many other areas of healthcare," explains Peter Pramstaller, Head of Eurac Research's Institute of Biomedicine.